Vaccines | Free Full-Text | Development and Evaluation of Vero Cell-Derived Master Donor Viruses for Influenza Pandemic Preparedness | HTML

Rabies vaccine purification using HPLC, Estimation of residual vero cellular DNA by RT-PCR and potency analysis | Madridge Publishers

GMP manufacturing of Vvax001, a therapeutic anti-HPV vaccine based on recombinant viral particles - ScienceDirect

Enhancing viral vaccine production using engineered knockout vero cell lines – A second look - ScienceDirect

Development of an Inactivated Vaccine Candidate, BBIBP-CorV, with Potent Protection against SARS-CoV-2 - ScienceDirect
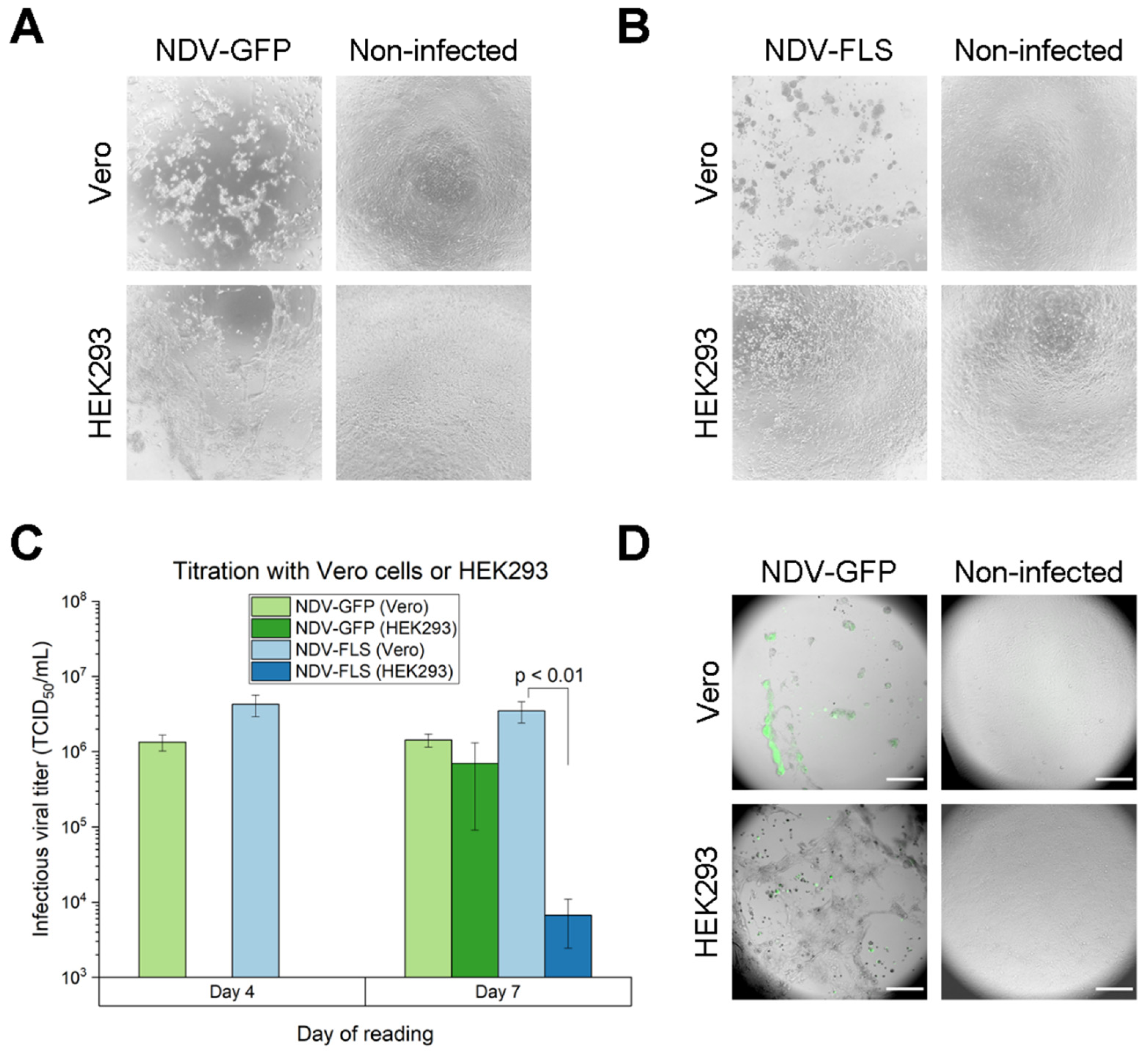
Vaccines | Free Full-Text | Process Development for Newcastle Disease Virus-Vectored Vaccines in Serum-Free Vero Cell Suspension Cultures
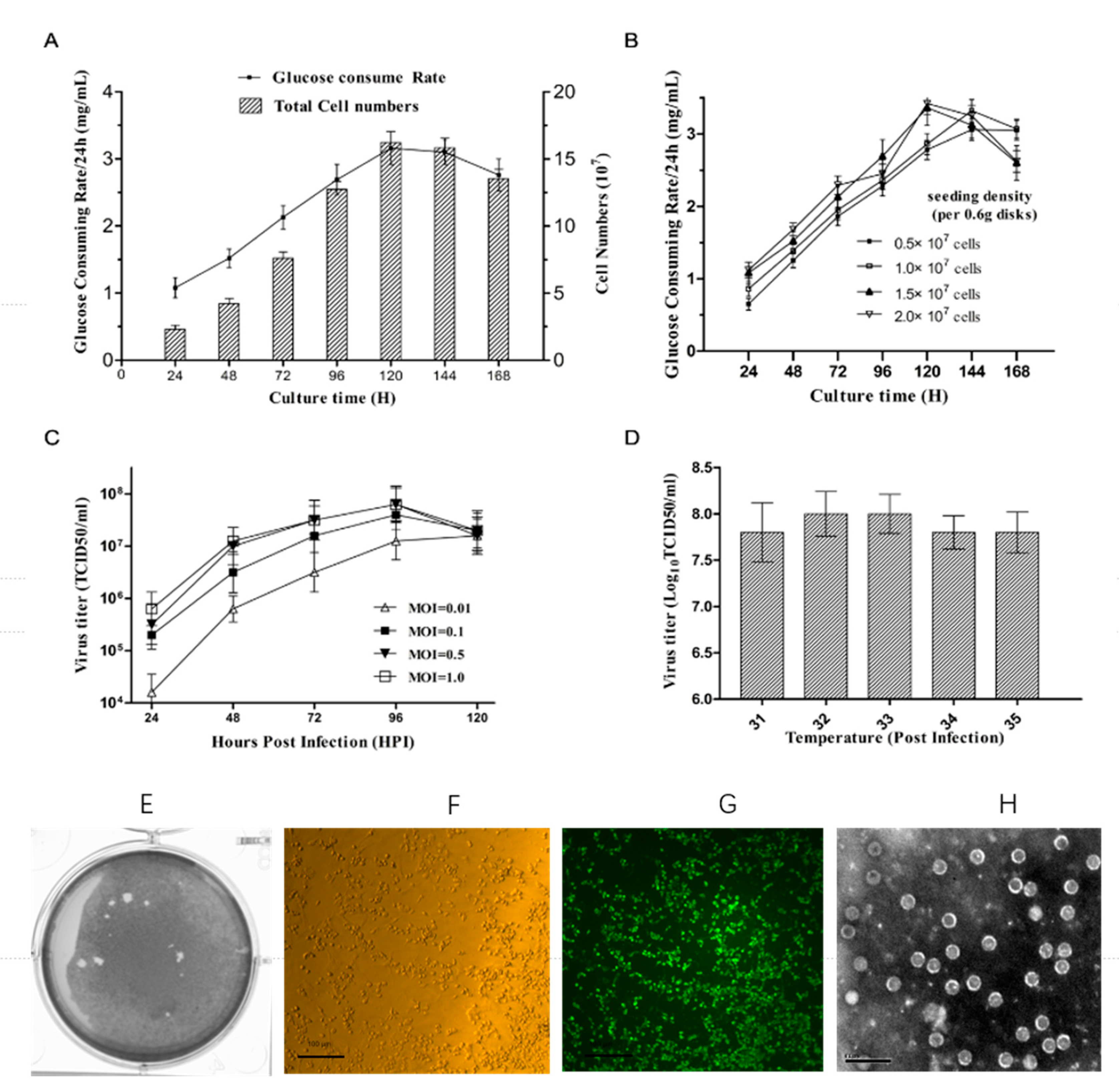
Vaccines | Free Full-Text | Optimization of Vero Cells Grown on a Polymer Fiber Carrier in a Disposable Bioreactor for Inactivated Coxsackievirus A16 Vaccine Development | HTML

Safety and immunogenicity of a Vero-cell-derived, inactivated Japanese encephalitis vaccine: a non-inferiority, phase III, randomised controlled trial - The Lancet

Impact Of A Chemically Defined Medium For Vero Cells Cultivation And Virus Production For Vaccine Applications

A whole virion vaccine for COVID-19 produced via a novel inactivation method: results from animal challenge model studies | bioRxiv

Efficacy, safety, and immunogenicity of a Vero-cell-culture-derived trivalent influenza vaccine: a multicentre, double-blind, randomised, placebo-controlled trial - The Lancet

![Sinopharm [Vero Cell]- Inactivated, COVID-19 vaccine Sinopharm [Vero Cell]- Inactivated, COVID-19 vaccine](https://www.who.int/images/default-source/departments/immunization-ivb/publications/sinopharm-vaccine-explainer-cover.tmb-479v.jpg?sfvrsn=28d56566_1)