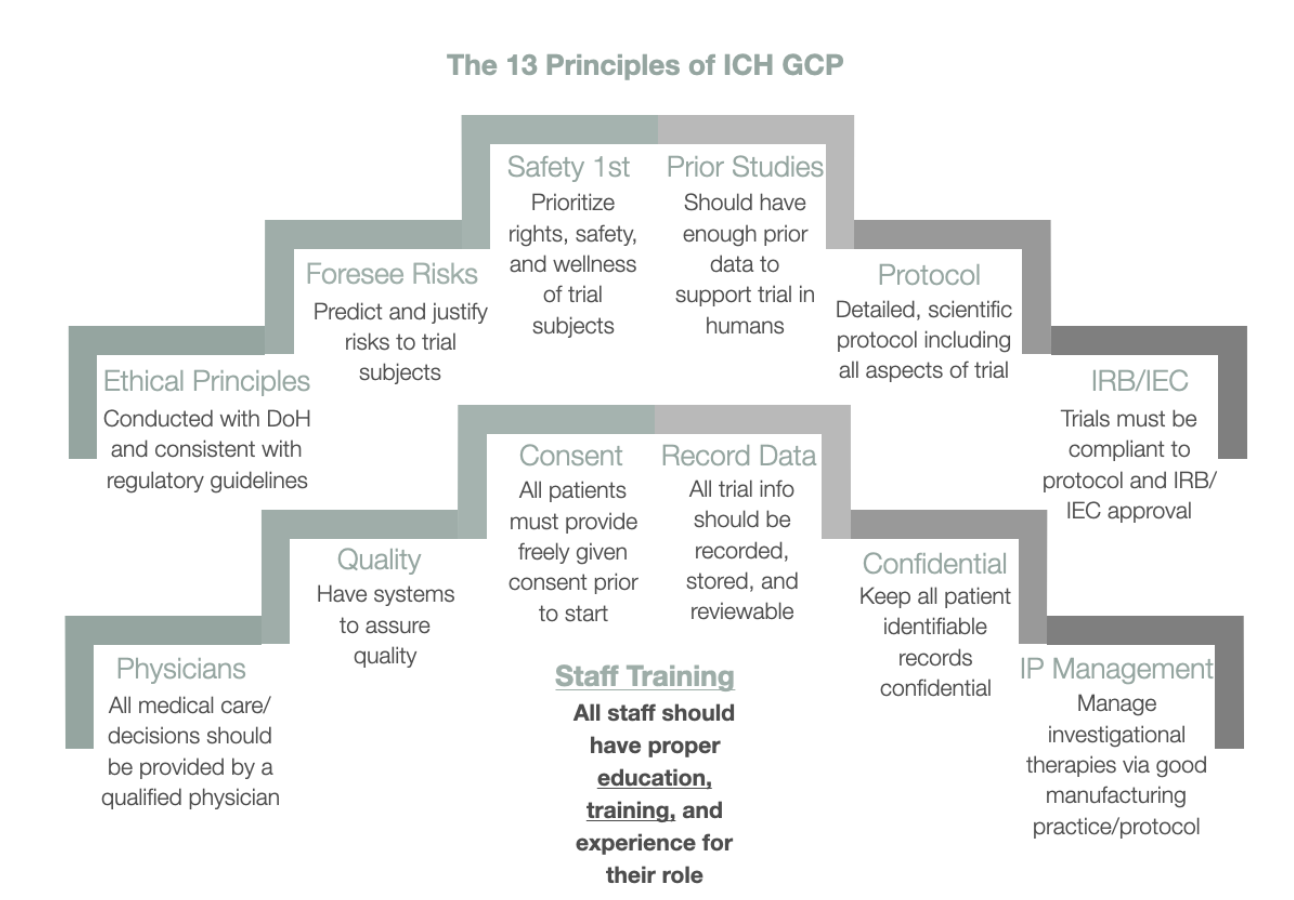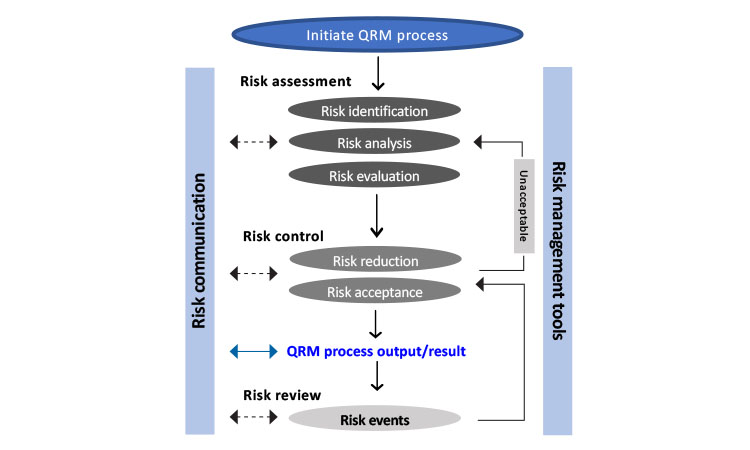
Clinical Trial Management Adaptation to ICH E6 (R2): Good Clinical Practice | Pharmaceutical Engineering

The Good Clinical Practice guideline and its interpretation – perceptions of clinical trial teams in sub‐Saharan Africa - Vischer - 2016 - Tropical Medicine & International Health - Wiley Online Library

Capacity Building - The Multi-Regional Clinical Trials Center of Brigham and Women's Hospital and Harvard